Due to its high absorption capacity, in Indonesia Microfiber has been widely used for various applications, for example; Car Wipe / Cloth Detailing:
There are many different brands of car wipes on the market. Its function is indeed different from the “Kanebo” Cloth or Chamois Cloth. “Kanebo” cloth are made of PVA and Microfiber is made of Polyester and Nylon.
Functionally and usability is the same for wiping or cleaning. But PVA can only be used when wet. Because if it’s dry, PVA will be hard like crackers. While Microfiber can be used when wet and dry. The advantages of Microfiber can be used for electronic items when dry. If PVA cannot be used for electronics because the cloth must always be wet, and if the cloth is wet it can cause electrical shorts and or damaged electronics/gadgets.
Wipe Kanebo PVA or Chamois
– What is meant by PVA?
Quoted from wikipedia:
”Polyvinyl acetate (PVA or PVAc ) is a synthetic rubber polymer. Polyvinyl acetate is made from its monomer, vinyl acetate (vinyl acetate monomer, VAM). This compound was discovered in Germany by Dr. Flitz Klatte in 1912. Complete or partial hydrolysis of this compound yields polyvinyl alcohol (PVOH). The yield ratio of this hydrolysis ranged from 87% – 99%.
PVA is sold as an emulsion in water, as an adhesive for porous materials, especially wood. PVA is the most commonly used wood glue, either as “white glue” or “carpenter’s glue” (yellow glue). The “yellow glue” is also widely used for gluing other materials such as paper, cloth and cigarettes. PVA is also commonly used in book printing because of its flexibility and is not acidic like many other polymers. Elmer glue is a well-known brand of PVA glue in the United States.
PVA is also often used as a copolymer with acrylate (which is more expensive), used in paper and paint. This copolymer is called vinyl acrylate. PVA can also be used to protect cheese from mold and moisture. PVA reacts slowly with bases to form acetic acid as a result of hydrolysis. Boron compounds such as boric acid or borax will form as a precipitate. “
While the microfiber cloth is 80% polyester, 20% Polyamide (Nylon).
– Polyester
Quote from wikipedia:
Polyesters are a category of polymers that contain an ester functional group in their main chain. Although there are many different types of polyesters, the term “polyester” as a specific material refers more often to polyethylene terephthalate (PET). Polyester includes natural chemicals, such as those extracted from the skins of plants, as well as synthetic chemicals such as polycarbonate and polybutyrate.
Can be produced in various shapes such as sheets and 3-dimensional shapes, polyester as a thermoplastic can change shape when heated. Although flammable at high temperatures, polyester tends to shrink away from fire and self-extinguishes when combustion occurs. Polyester fiber has high strength and E-modulus as well as low water absorption and minimal shrinkage when compared to other industrial fibers.
Woven polyester fabrics are used in consumer clothing and home furnishings such as bed sheets, bed covers, curtains and drapes. Industrial polyesters are used in tires, ropes, fabrics for conveyor belts, seat belts, quilted fabrics and plastic reinforcements with a high energy absorption rate. Polyester fiber fill is also used to fill pillows and warm blankets.
Fabrics made of polyester are said to feel “unnatural” when compared to similarly woven fabrics of natural fibers (eg cotton in textile use). However, polyester fabrics have several advantages such as increased resistance to shrinkage. As a result, polyester fibers are sometimes spun together with natural fibers to produce garments with combined properties.
Polyesters are also used to make bottles, films, tarpaulin, canoes, liquid crystal displays, holograms, filters, dielectric film for capacitors, insulating film for cables and insulating tape.
Liquid crystalline polyester is one of the first industrially used liquid crystalline polymers and is used because of its mechanical properties and resistance to heat. This advantage is important in its use as an aerodible seal in jet engines.
Thermosetting polyester is used as a casting material, and chemosetting polyester resin is used as a glass fiber coating resin and non-metallic car body putty. Fiberglass-reinforced unsaturated polyesters are widely used in the bodies of yachts and cars.
Polyester is also widely used as a finish in high-quality wood products such as guitars, pianos, and vehicle/yacht interiors. Burns London, Rolls-Royce and Sunseeker are among the few companies that use polyester to refine their products. The thixotropic properties of the sprayable polyester make it ideal for use on open-grain logs, as it is capable of filling wood grain quickly, with a strong film thickness forming per layer. The cured polyester can be sanded and polished to the final product.
PROPERTIES OF POLYESTER FIBER
Mechanical properties
Energy absorption of chemical fiber reinforced plastics (impact, bending and tensile tests) Investigation of the practical requirements for measuring the energy absorption of composite materials (composite), and development of suitable methods for carrying out such measurements. A number of dynamic test methods to measure the energy absorption of various layers, including bending impact test, repeated impact test, tensile impact test and bending impact test.
Also discussed is the impact test on plated slabs. Particular emphasis is placed on the study of various composites reinforced with a chemical fiber. It is undeniable that there is a relationship between the apparent static energy absorption of the various fibers and the dynamic energy absorption of the composites. Commercial polyester composites and polyamide fibers have the highest energy absorption, on which the test apparatus has a significant effect.
Chemical nature
Polyester is not known to have chemical properties.
POLYESTER INDUSTRY
Basics
Polyester is a synthetic polymer made of Purified Terephthalic Acid (PTA) or dimethyl ester dimethyl terephthalate (DMT) and Mono Ethylene Glycol (MEG). With a market share of 18% of all plastic materials produced, polyester ranks third after polyethylene (33.5%) and polypropylene (19.5%).
The main raw materials are as follows:
- Purified Terephthalic Acid – PTA – CAS-No.: 100-21-0
Synonyms: 1,4 Dibenzenedicarboxylic acid,
Sum formula; C6H4(COOH)2 , berat mol: 166,13
- Dimethylterephthalate – DMT- CAS-No: 120-61-6
Synonyms: 1,4 Dibenzenedicarboxylic acid dimethyl ester
Sum formula C6H4(COOCH3)2 , berat mol: 194,19
- Mono Etilena Glikol – MEG – CAS No.: 107-21-1
Synonyms: 1,2 Ethanediol
Sum formula: C2H6O2 , berat mol: 62,07
More information on various polyester raw materials can be found for PTA [1], DMT [2] and MEG [3], on the INCHEM website “Chemical Safety Information from Intergovernmental Organizations”.
A catalyst is needed to produce a polymer with a high molecular weight. The most commonly used catalysts are antimony trioxide (or antimony tri acetate):
Antimony trioxide – ATO – CAS-No.: 1309-64-4 Synonyms: none, mole weight: 291.51 Sum formula: Sb2O3
In 2008, about 10 000 t of Sb2O3 were used to produce about 49 Mio t of polyethylene terephthalate.
Polyester is described as follows:
Polyethylene Terephthalate CAS-No.: 25038-59-9 Synonyms / abbreviations: polyester, PET, PES Sum Formula: H-[C10H8O4]-n=60-120 OH, unit mole weight: 192.17
There are several reasons why PTA is important:
- Relatively easy access to various raw materials PTA or DMT and MEG
- The chemical process is polyester synthesis which is easy to explain and very easy to understand
- Low toxicity of all raw materials and by-products during production and processing
- PET can be produced in a closed loop at low emissions to the environment
- Can be recycled
- Many variants of intermediate and final products made of polyester
- In table 1: world production of polyesters for textile polyesters, bottle polyester resins, film polyesters especially for packaging and specialty polyesters for machine plastics. According to this table, world polyester production exceeded 50 million tonnes annually before 2010.
| Market Share per Year | ||
| Product Type | 2002 [Mio t/a] | 2008 [Mio t/a] |
| Txstil -PET | 20 | 39 |
| Resin, Bottle/A-PET | 9 | 16 |
| Film-PET | 1.2 | 1.5 |
| Special Polyester | 1 | 2.5 |
| TOTAL | 31.2 | 49 |
Raw material manufacturers
Most of the PTA, DMT and MEG raw materials produced by large chemical companies are sometimes integrated into crude oil refining where p-xylene is the base material to produce PTA and LPG is the base material for producing MEG.
BP, Reliance, Sinopec, SK-Chemicals, Mitsui, and Eastman Chemicals are examples of the many PTA manufacturers. MEG production is in the hands of around 10 global players led by MEGlobal a JV of DOW and PIC Kuweit followed by Sabic.
The following are the names of the largest polyester manufacturers:
Artenius, Advansa, DAK, DuPont, Eastman/Voridian, Hyosung, Huvis, Indorama, Invista, Jiangsu Hengli Chemical Fiber, Jiangsu Sanfangxian Industry, M&G Group, Mitsui, Mitsubishi, NanYa Plastics, Reichhold, Reliance, Rongsheng, Sabic, Teijin, Toray , Trevira, Tuntex, Wellman, Yizheng Sinopec, Zhejiang Hengi Polymerization.
There are more than 500 polyester factories in China, so it’s no wonder that half of the world’s polyester production comes from that country. More information about polyester in China can be found on the China Chemical Fiber Economic Information Network website.
Polyester processing
After the first stage of polymer production in the melt phase, the product stream is divided into two distinct application areas namely textile applications and packaging applications. Table 2 lists the main applications of packaging and textile polyesters.
| Textil | Packing |
| Stapel Fiber (PSF) | Bottle untuk CSD, Air, Bir, Jus, Deterjen |
| Filamen POY, DTY, FDY | A-PET Film |
| Technical thread and tire wire | Thermoforming |
| Non-woven and spunbond | BO-PET |
| Mono-filamen | Dressing |
Abbreviation: PSF = Polyester Staple Fiber; POY = Partially Oriented Yarn; DTY = Draw Textured Yarn; FDY = Fully Drawn Yarn; CSD = Carbonated Soft Drink (soft drink filled with carbon gas); A-PET = Amorphous Polyester Film (shapeless polyester film); BO-PET = Biaxial Oriented Polyester Film (biaxially oriented polyester film);
Polyester’s small market share (<< 1 Million t/a) is used to produce technical plastics and master packaging.
In order to produce melt polyester very efficiently, some high-output processing steps such as staple fiber (50–300 t/s per spinning line) or POY /FDY (up to 600 t/s separated into about 10 spinning machines) are increasingly horizontal processes , integrated, and direct. This means that the melted polymer is directly converted into textile filaments or fibers without going through a granulation step.
We are discussing fully horizontal integration when polyester is produced from crude oil or various refined products in chain oil -> benzene -> PX -> PTA -> melted PET -> fiber/filament or bottle-grade resin. Eastman Chemicals was the first to introduce the idea of chain sealing from PX to PET resins with what they call the INTEGREX® process. The capacity of such an integrated horizontal production site is >1000 t/d and can easily reach 2500 t/d.
In addition to the large processing units for producing staple yarns or fibres, there are tens of thousands of small and very small processing plants, so it can be estimated that polyester is processed and recycled in more than 10,000 factories worldwide. This is without counting all the companies involved in the supply chain industry, starting with engineering and processing machinery and ending with additional stabilizers, colors and additives.
Synthetic
The synthesis of polyester is generally achieved by a polycondensation reaction. The general formula for the reaction of a diol with a dicarboxylic acid is:
(n+1) R(OH)2 + n R´(COOH)2 —> HO[ROOCR´COO]nROH + 2n H2O
Azeotropic esterification
In this classical method, one alcohol and one alkanoic acid react to form a carboxylic ester. To assemble a polymer, the water formed from the reaction must be continuously removed by azeotropic distillation.
Alcoholic transesterification
| O \\ C – OCH3 + OH[Oligomer2] / [Oligomer1] | 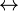 | O \\ C – O[Oligomer2] + CH3OH / [Oligomer1] |
| (ester-terminated oligomer + alcohol-terminated oligomer) | (larger oligomer + methanol) |
Acylation (HCl method)
The acid begins as a hydrochloric acid, and so the polycondensation continues to emit hydrochloric acid (HCl), instead of water. This method can be done in solution or as an email.
Silyl method
In this variant of the HCl method, the alkanoic acid chloride is converted with the trimethyl silyl ether of the alcohol component and the result is trimethyl silyl chloride.
Ring-opening polymerization
Aliphatic polyesters can be prepared from lactones at room temperature and 1 atm pressure, catalyzed by anions, cations, or organometallic (metalorganic).
THERMOSETTING (HOT)
In general, thermosetting resins are copolymers of unsaturated polyesters with styrene. The saturation of the polyester is regulated through the use of both maleic acid and fumaric acid. In vinylester, the saturation is in the alcohol group of the polyester. The double bond unsaturated polyester reacts with styrene and produces a 3-D crosslinked structure. This structure acts as a thermoset. Cross-linking is initiated by an exothermic reaction involving an organic peroxide, such as methyl ethyl ketone peroxide or benzoyl peroxide.
Polyamide (Nylon)
Nylon is a family of synthetic polymers created in 1935 by Wallace Carothers at DuPont. The first product was a nylon bristle toothbrush (1938), followed by a better known product: stockings for women in 1940. Nylon is made from a series of units linked by peptide bonds (amide bonds) and is often referred to as polyamide (PA). Nylon was the first commercially successful polymer, and the first synthetic fiber made entirely from inorganic materials: coal, water and air. These elements are arranged into low molecular weight monomers, which are then reacted to form long polymer chains.
This material was intended to be a synthetic substitute for silk which was realized by using it to replace silk as parachute material after the United States entered World War II in 1941, which made stockings difficult to obtain until the war ended.
Nylon fibers are now used for fabrics and ropes. Solid nylon is used for mechanical and engineering parts





